Important updates for Turkey KKDIK registrants
The Turkish Ministry of Environment and Urbanization and Climate Change – MoEUCC – communicated last week important news regarding the sections 1.7. Suppliers and 13.1 Chemical Safety Report of the KKDIK registration dossiers as follows:
Updates to section 1.7: Suppliers
The MoEUCC has taken a closer look at the opinions and recommendations expressed by industry associations, institutions, and various chemical manufacturing companies in relation to the section 1.7 of the registration dossiers where Only Representatives (ORs) are required to include in the dossier information of the importers in Turkey of the overseas companies they represent. After further consideration the MoEUCC has decided yesterday that this requirement will be optional but not mandatory. In other words, if the appointed ORs don’t include such information under section 1.7 of their dossiers, it won’t be an obstacle for passing the completeness checks as it won’t be considered as missing information.
However, this update won’t exclude the responsibility for appointed ORs of keeping up-to-date information of the importers in Turkey of the Non-Turkish manufacturer/formulator they represent including substances annual tonnage and information on how to provide updated Safety Data Sheets. In case of inspections the ORs are required to show this information to the authority.
1.3. Chemical Safety Report – Updates
A chemical safety report (CSR) is an essential part of the KKDIK registration dossier and must be added to the registration dossier upon submission to the MoEUCC. The MoEUCC has announced that KKDIK registrants are allowed to upload the CSR in English language. This new approach will help registrants to expedite the registration of their substances so that they are able to meet the registration deadline on time (31.12.2023). However, please note that KKDIK registrants who choose to upload a Chemical Safety Report in English are anyways required to include a Turkish translation of the full CSR, which includes the uses, exposure scenarios and risk management measures in Turkey, at the latest on 31st of December 2024.
Authority Fees – Updated
In January, 2023 the MoEUCC has published updated authority fees. Take a look at the fees you will need to pay to the MoEUCC as part of the KKDIK registration process for your substances. (More information on the fees can be found here: https://donersermaye.csb.gov.tr/ )
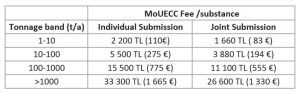
*Exchange rate ( TL to EUR) of 9th February, 2023)
Need Support?
Via our office in Istanbul we would be glad to supporting your company as your Only Representative or service provider to prepare and submit your registrations to the MoEUCC by the 31st of December, 2023 deadline. Contact us at sales@reachlaw.fi for support.
Never miss a story
Stay updated about REACHLaw Blog as it happens


