1 January 2024: Poison Centre Notification deadline for industrial use mixtures
Background Information:
Article 45 of CLP requires importers and downstream users to notify hazardous mixtures to the appointed bodies (i.e,
poison centers) using the harmonized format (Annex VIII) following deadlines for each use category.
With deadlines passed for Professional and consumer use, Industrial use mixtures are required to be notified following
deadline of 1st Jan 2024.
Decide if your business is affected by the 2024 deadline:
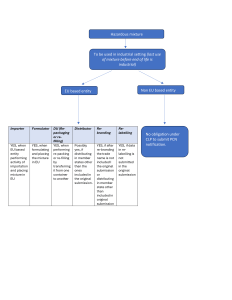
Confidentiality provisions in PCN:
Limited submission: Limited submission is an option applicable only for Industrial use mixtures. Limited submission
allows companies to maintain information related to composition/ formula where composition referring to Section 3 of
SDS is enough. However, full composition has to be submitted if the use extends to consumer or professional category.
Role of Non-EU manufacturers/formulators in the notification process:
Non – EU entity never has the obligation under CLP to perform the PCN notification. However, they can provide
information to EU based DU/ importer to perform the notification.
In case non-EU based entity wants to keep the information confidential, one way is to assign a EU based entity to perform
voluntary submission by generating a UFI number. Further EU based entity makes their own submission with reference to this UFI number in relation to the compositional information.
Tips to consider when preparing your PCNs:
– Decide correct use category as per end use of your hazardous mixture.
– Decide your role accurately.
– Submission of data with accuracy. In case of limited submission, SDSs should be compliant with CLP.
– Manage UFI number smartly.
– Act as per deadline
For further information, do not hesitate in contact us at sales@reachlaw.fi .
Never miss a story
Stay updated about REACHLaw Blog as it happens


